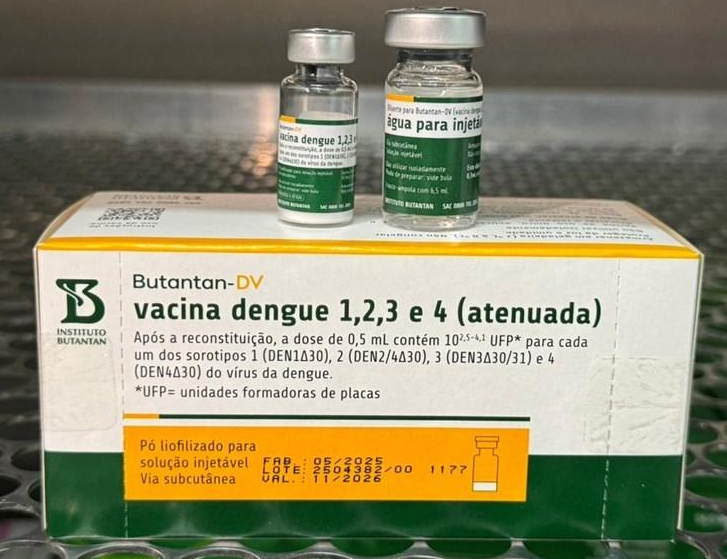
Dengue vaccine 1, 2, 3, and 4, live attenuated
| Product Overview | |
| Generic Name | Dengue vaccine 1, 2, 3, and 4, live attenuated |
| Brand Name(s) | BUTANTAN-DV™ |
| Form | Vial, Sub-C Inject |
| Strength | Single Dose |
| Therapeutic Class | Vaccines; viral vaccines |
| ATC Code | J07BX04 |
| Manufacturing & Regulatory | |
| Manufacturer | Instituto Butantan, São Paulo, Brazil |
| Country | Brazil |
| GMP Compliance | WHO-GMP |
| COFEPRIS | Pending |
| Free Sale Certificate | Prescription use; sale prohibited to commerc |
| Logistics & Export | |
| MOQ | 10,000 Single Dose Vials |
| Shelf Life | 18 months |
| Incoterms | EXW São Paulo, Brazil |
| Lead Time | 45 days |
| Documentation | |
| Certificate of Analysis (COA) | Yes—lot-specific Certificate of Analysis |
Description
Butantan professional package insert lists the product as Butantan-DV™ vacina dengue 1, 2, 3 e 4 (atenuada), registered and produced by Instituto Butantan, with registration number 1.2234.0057, and states that the package insert was approved by ANVISA on 08/12/2025