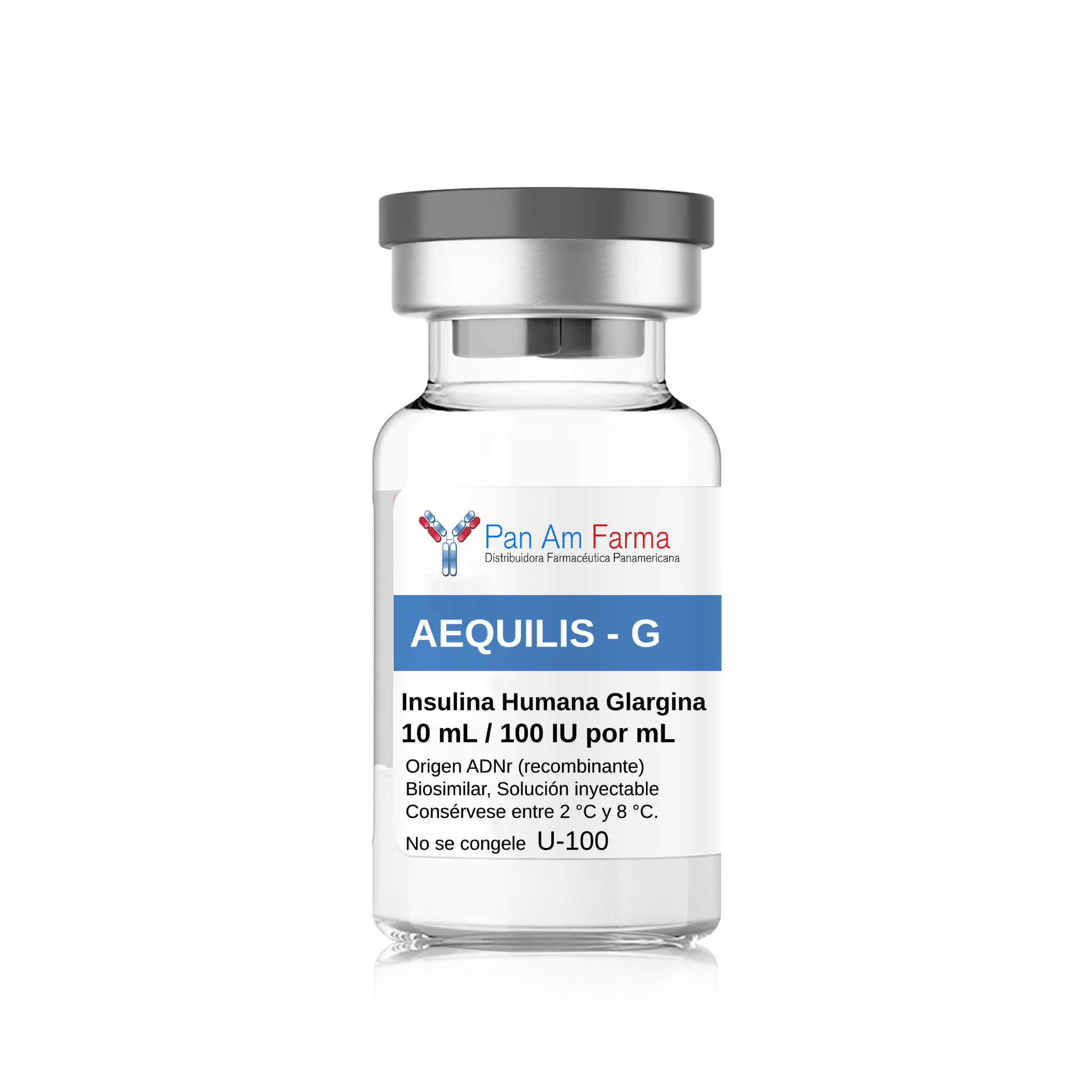
Insulin Glargine Biosimilar 10ml
| Product Overview | |
| Generic Name | Insulin Glargine Biosimilar 10ml |
| Brand Name(s) | AEQUILIS - G |
| Form | Vial |
| Strength | 10mL / 100 IU por mL |
| Therapeutic Class | Antidiabetic agent; long‑acting insulin analogue |
| ATC Code | A10AE04 |
| Manufacturing & Regulatory | |
| Manufacturer | PanAmFarma |
| Country | Mexico |
| GMP Compliance | WHO-GMP, EU-GMP |
| COFEPRIS | Pending Registration |
| Free Sale Certificate | Available per batch upon request |
| Logistics & Export | |
| MOQ | 440 vials |
| Shelf Life | 24 Months |
| Storage | 2–8 °C in fridge; do not freeze; |
| Incoterms | Ex-Works Mexico |
| Lead Time | 7 to 10 Days |
| Documentation | |
| Certificate of Analysis (COA) | Manufacturer provides COA |
| SDS | Available from manufacturer |
| CTD Summary | Available under confidentiality; summary upon significant purchase |
Description
AEQUILIS G, Insulina Glargina, Origen ADNr, Solución inyectable 100 UI/mL. Frasco ámpula 10 mL / Contenido total: 1,000 UI. Vía subcutánea. No administrar por vía intravenosa. No mezclar. No diluir. Consérvese entre 2 °C y 8 °C. No se congele.