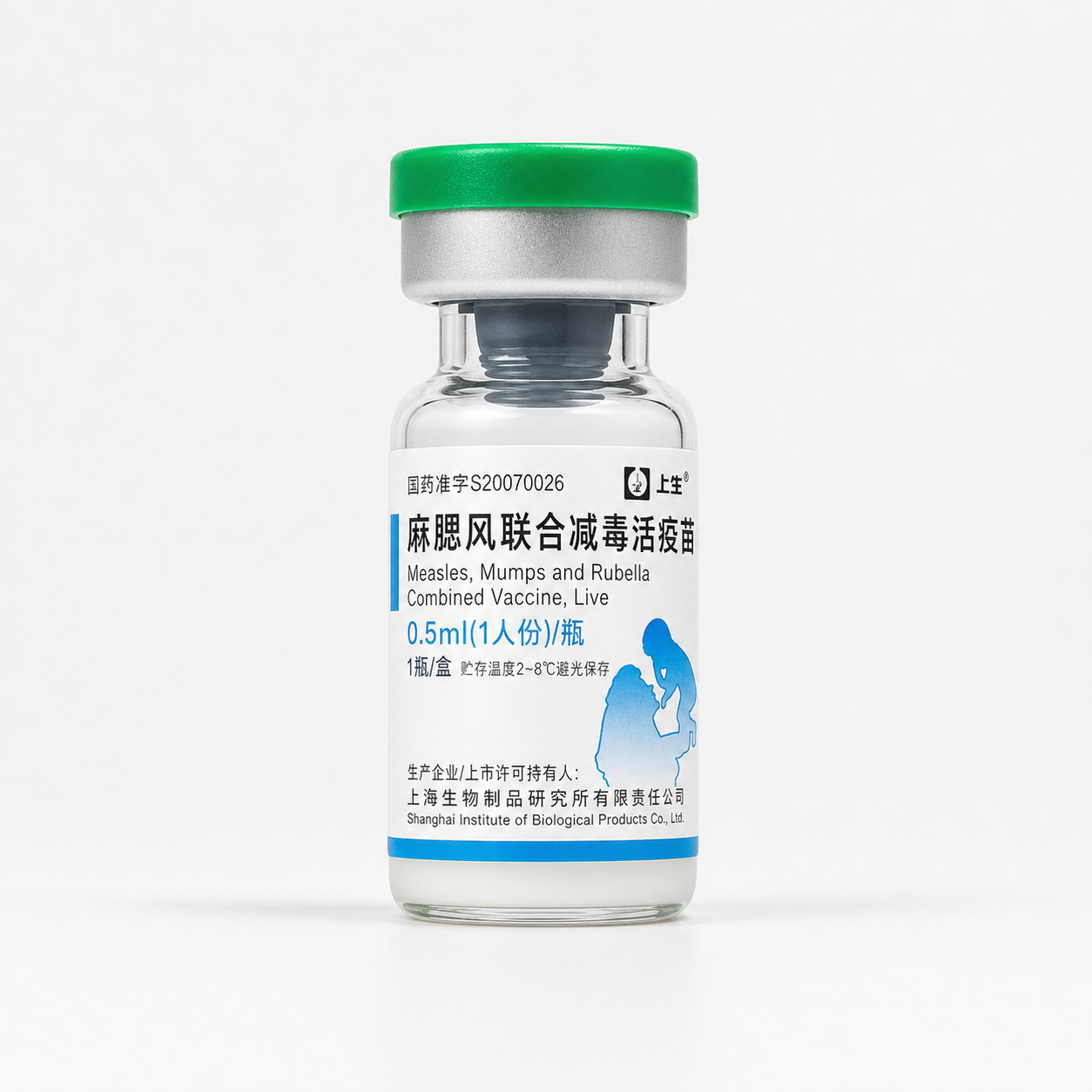
MMR, Measles-Mumps-Rubella Vaccine
| Product Overview | |
| Generic Name | MMR, Measles-Mumps-Rubella Vaccine |
| Brand Name(s) | SIBP MMR Vaccine |
| Form | 0.5mL Single Dose Vial |
| Strength | 0.5 mL after reconstitution |
| Therapeutic Class | Viral vaccine; live attenuated combination vaccine |
| ATC Code | J07BD52 |
| Manufacturing & Regulatory | |
| Manufacturer | Shanghai Institute of Biological Products Co., Ltd |
| Country | Shanghai, People’s Republic of China. |
| GMP Compliance | WHO-GMP |
| DMF/CEP | No public DMF number found |
| COFEPRIS | 020.000.3820.00 |
| Free Sale Certificate | For Latin America, request CPP, FSC |
| Logistics & Export | |
| MOQ | 10,000 |
| Shelf Life | 18 - 24 Months |
| Storage | 2–8 °C in fridge; do not freeze; |
| Incoterms | EXW |
| Lead Time | 4–8 weeks |
| Documentation | |
| Certificate of Analysis (COA) | Batch-release certificate, lot-specific cold-chain release statement |
| SDS | No public SDS found for this exact SIBP product |
| CTD Summary | request a vaccine CTD / ACTD / eCTD summary package |
Description
Measles, Mumps and Rubella Combined Vaccine,live attenuated biological vaccine and must be handled under validated cold-chain conditions